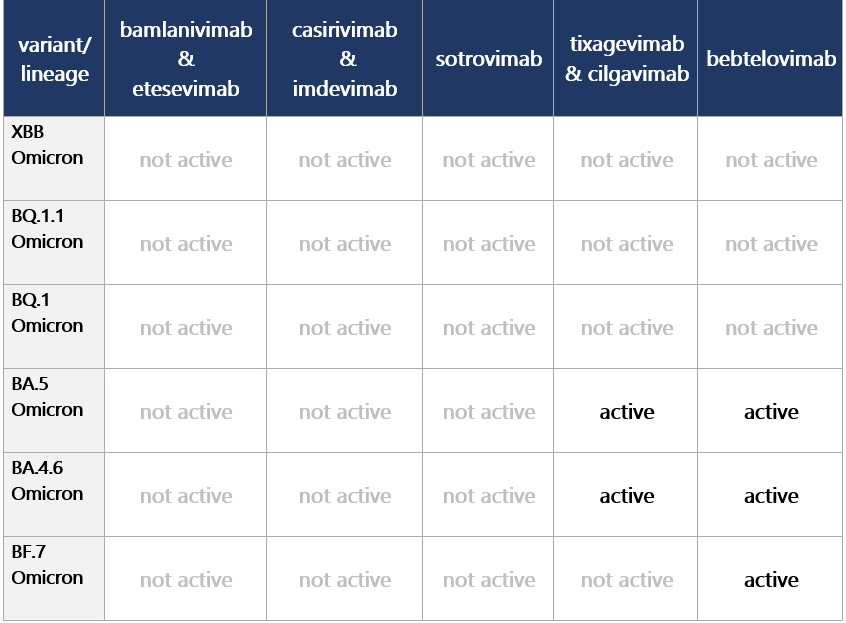
No monoclonal antibody currently authorized for COVID-19 treatment or prophylaxis in any U.S. region d/t high frequency of non-susceptible variants.
Step 1: Check CDC genomic surveillance site(s) for variant prevalence.
Step 2: Check Monoclonal Antibody Activity to determine optimal monoclonal antibody tx.
| Guideline | Resources |
| SARS-CoV-2 Variant-Based Monoclonal Antibody Selection Tool |
Key Points
Steps
|
This epocrates resource is developed by our in-house team of pharmacist and physician editors.
Epocrates Guideline Synopsis Last Update:
Jan 27, 2023
Publication Year:
2022
Source:
epocrates